Human prekallikrein
Freeze-dried

Packaging options
| 26-ADG472 | Vial | 1 mg |
Description of the Human prekallikrein
Sodium acetate, 0.15 M sodium chloride, pH 5.3.
MW(Da) : 86 000
Extinction coef. : 11.7
Purity > 95%
Informations
Prekallikrein (PK), also known as Fletcher factor, is a serine protease that complexes with high molecular-weight kininogen.
Prekallikrein is the zymogen form of plasma kallikrein, which is a serine protease that activates kinins. It is cleaved to produce kallikrein by activated FXII (Hageman factor).


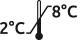

product sheet








