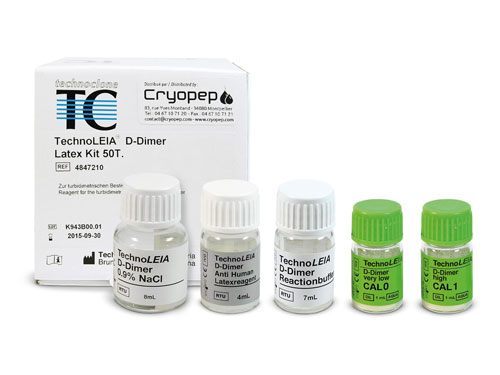
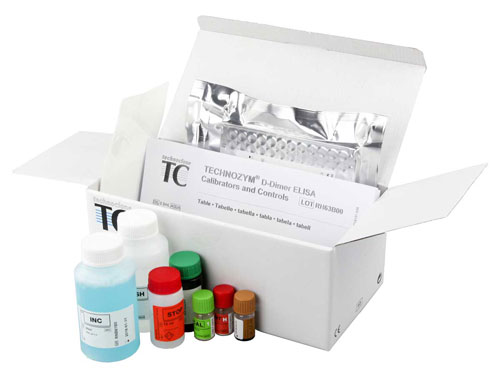
HEMOSTASIS COAGULATION ROUTINE REAGENTS D-DIMERS ELISA
TECHNOLEIA® D-Dimer Calibrator 0 ng/mL
Low D-Dimer calibration plasmas
0 ng / mL calibration plasmas used for the Technoleia® D-Dimer assay kit.
Informations
D-Dimers are fragments resulting from the destruction of fibrin during fibrinolysis.
A low level of D-Dimer is normal and indicates that there has been activation of coagulation and formation of a clot, but can sharply increase in cases of venous thrombosis, DIC (disseminated intravascular coagulation) or pulmonary embolism.
Documentation
Download the product sheetPrice list, safety data sheets and notices are accessible to our registered customers.

Immuno-Latex assays








References
| 4-4847236 | Vial | 2 x 1.0 mL |