HEMOSTASIS COAGULATION ROUTINE INSTRUMENTS T-TAS®01 CONSUMABLES HD CHIPS - T-TAS® 01
Anti-sedimentation reagent for HD Chip T-TAS®01


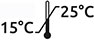


Reagent to prevent sedimentation of samples that will be tested using the HD chip for T-TAS®01.

| Reference | 25-NS0001 |
|---|---|
| Presentation | Vial |
| Format | 1 x 2.0 mL |
| Quote |
Price list, safety data sheets and notices are accessible to our registered customers.
| Reference | Presentation | Format | Quote | Product sheet |
|---|---|---|---|---|
| 25-NS0001 | Vial | 1 x 2.0 mL |