HEMOSTASIS COAGULATION RESEARCH REAGENTS HUMAN PLASMAS GENETIC DISORDERS
Plasma from heterozygous prothrombin gene mutation G20210A
Advantages
Minimize test time.
Ready to use.
Informations
The polymorphism of the prothrombin gene associated with a moderate hereditary thrombophilia corresponds to the replacement of a guanine by an adenine at position 20210, (3 'non-coding region of the prothrombin gene). The prothrombin level is on average higher.
The frequency of this gene variation is 2% in the general population of Caucasian origin and 7% in patients with a history of venous thromboembolism.
Documentation
Download the product sheetPrice list, safety data sheets and notices are accessible to our registered customers.



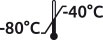


References
| 6-PPMFIIHE | Vial | 1 x 1.0 mL |