HEMOSTASIS COAGULATION RESEARCH REAGENTS DEFICIENT PLASMAS CONGENITAL DEFICIENT PLASMAS (BOTTLES)
Human Factor XII congenital deficicent plasma >5%
Plasma from a human donor with congenital FXII deficiency.
Packaging in bottle. The minimum packaged volume is 50 mL. The price offer is based on the volume requested.
Advantages
Minimize test time.
Ready to use.
Informations
Factor XII (FXII) is a glycoprotein synthesized in the evening.
FXII participates in the contact phase which initiates the intrinsic pathway of coagulation.
Activated on contact with a negatively charged surface, it becomes capable of activating prekallikrein and kallikrein (amplified by KHPM) then FXI to FXIa in the presence of KHPM.
The FXIa thus formed activates the FXII in FXIIa, amplifying the reaction.
Documentation
Download the product sheetPrice list, safety data sheets and notices are accessible to our registered customers.



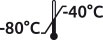


References
| 6-PPD12C | Vial | Minimum 50 mL |