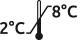HEMOSTASIS COAGULATION RESEARCH REAGENTS PLASMA DERIVED PROTEINS TISSUE FACTOR
Recombinant tissue Factor
Formulation : 20 mM Tris, 150 mM NaCl, 10 mM CHAPS, pH 8.0
MW(Da) : 35 000 Extinction coef. : 12.6
Advantages
The vast majority of plasma derivatives is pure (without additives) with > 95 % purity SDS-PAGE. Expiration date of one year from delivery. Delivery in large quantities. Discount according to quantities.
Informations
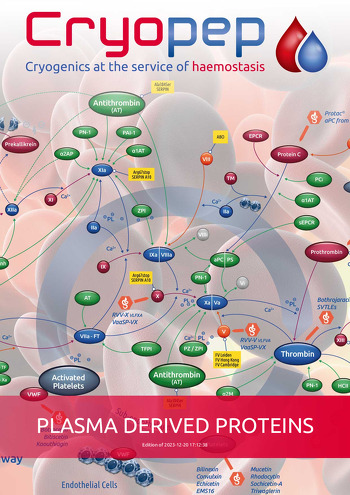
Tissue factor (FT) is a transmembrane glycoprotein which is primarily responsible for activating coagulation cascades in the event of a vascular breach. The binding of FVII to its receptor, expressed by the cells of the subendothelium exposed by the lesion, allows its very rapid activation by traces of FXa, circulating in trace amounts in vivo. The FT-FVIIa complex then causes the activation of FIX and FX and the formation of thrombin.
Documentation
Download the product sheetPrice list, safety data sheets and notices are accessible to our registered customers.


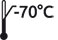


References
| 9-RTF-0300 | Vial | 10 µg |