HEMOSTASIS COAGULATION ROUTINE REAGENTS SCREENING TESTS PT APTT FIBRINOGEN TT
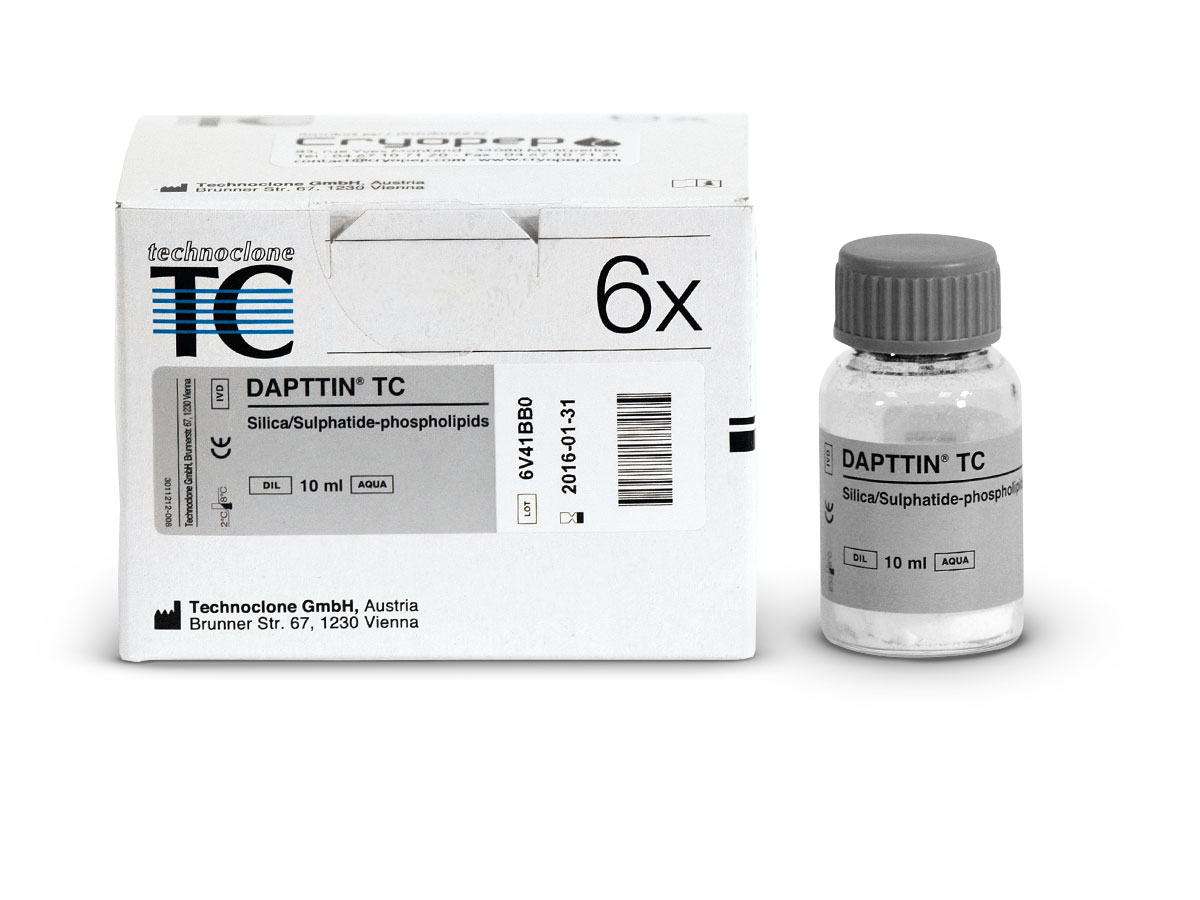
DAPTTIN® TC
TCA reagent to detect deficiencies of coagulation factors II, V, VIII, IX, X, XI and XII, lupus anticoagulants and to monitor patients during treatment with heparin.
Dapttin® TC reagent (2-activator cephalin) is a reactive for activated partial thromboplastin time (TCA) standardized in hemostasis, composed of 2 surface activators : kaolin and sulfatide, and a mixture of highly purified phospholipids.
This routine test is distinguished by an optimized behavior with regard to all coagulation factors and inhibitors.
Documentation
Download the product sheetPrice list, safety data sheets and notices are accessible to our registered customers.
Chronometric assay









References
| 4-5035060 | Vial | 5 x 2.0 mL |
| 4-5035090 | Vial | 6 x 10.0 mL |
| 4-5035100 | Vial | 20 x 10.0 mL |