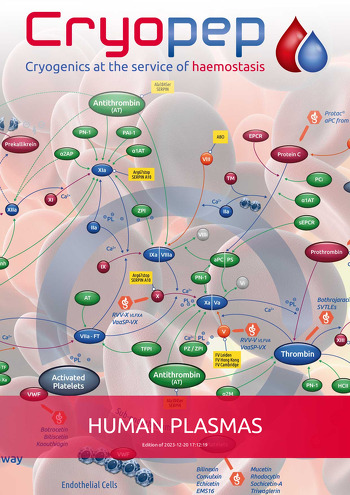
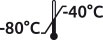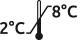Factor II (FII) is a glycoprotein synthesized by the liver, zymogen of a serine protease. It is a vitamin K-dependent clotting factor. Its half-life is 50 to 120 hours. FII is activated by the prothrombinase thrombin complex which plays a central role in the coagulation process. It will transform fibrinogen into fibrin, amplify its own formation and activate the protein C, TAFI and platelet systems. There are constitutional deficits in FII which are very rare and acquired deficits which can be observed during anti-vitamin K treatment or vitamin K deficiency, CIVD, anti-FII autoantibodies.
Special plasmas are derived from patients with acquired deficiency, severe or moderate, or presenting a particular profile. No buffer or preservatives are added. Quickly frozen at -80° C, the plasma maintains perfectly intact the matrix. All plasmas are stable when stored at -40° C to -80° C. We carefully pack with dry ice during shipment. No additive or preservative. Expiry date > 1 year. Plastic vials.
Minimize test time. Ready to use.