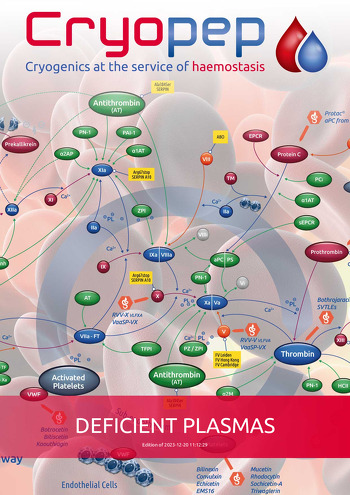
HEMOSTASIS COAGULATION RESEARCH DEFICIENT PLASMAS IMMUNODEPLETED DEFICIENT PLASMAS
Plasma Factor VIII deficient chemically depleted





Plasma deficient for the determination of Factor VIII.
| Reference | 9-FVIII-CD |
|---|---|
| Presentation | Vial |
| Format | from 50 mL |
| Quote |
Price list, safety data sheets and notices are accessible to our registered customers.
| Reference | Presentation | Format | Quote | Product sheet |
|---|---|---|---|---|
| 9-FVIII-CD | Vial | from 50 mL |