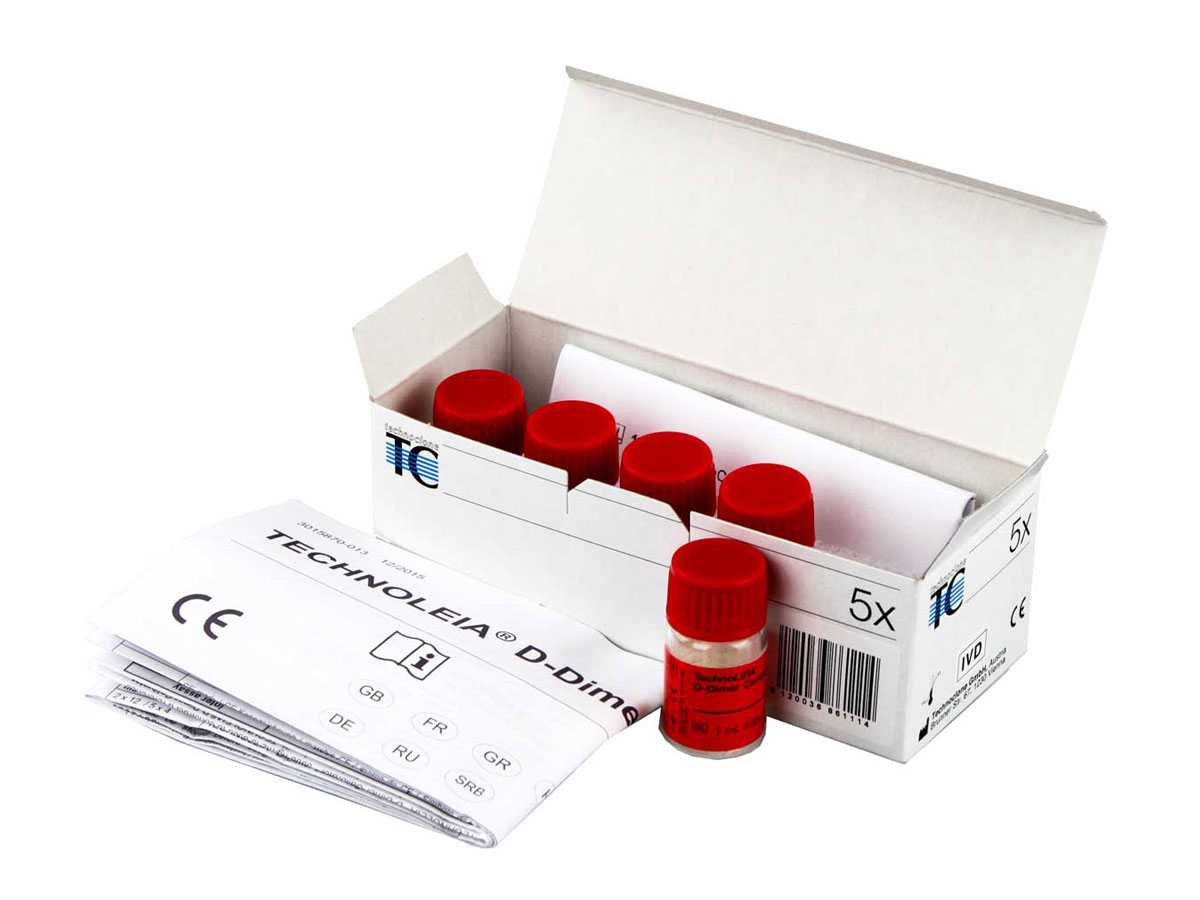
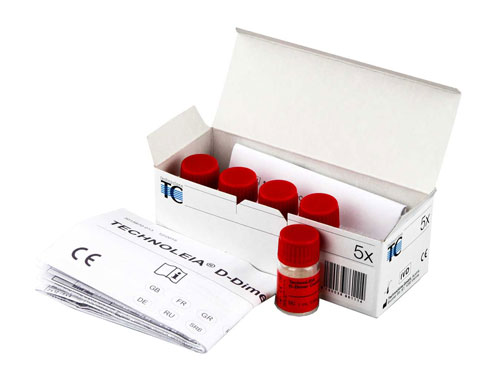
TECHNOLEIA® D-Dimer Control Low








Immuno-Latex assays
Low D-Dimer control plasmas
300 ng / mL control plasmas used for the Technoleia® D-Dimer Latex kit assay kit.
Access on the manufacturer's website TECHNOCLONE
| Reference | 4-4847232 |
|---|---|
| Presentation | Vial |
| Format | 5 x 1.0 mL |
| Quote |
Price list, safety data sheets and notices are accessible to our registered customers.
| Reference | Presentation | Format | Quote | Product sheet |
|---|---|---|---|---|
| 4-4847232 | Vial | 5 x 1.0 mL |