HEMOSTASIS COAGULATION ROUTINE FROZEN CALIBRATORS AND CONTROLS SCREENING TEST CONTROLS
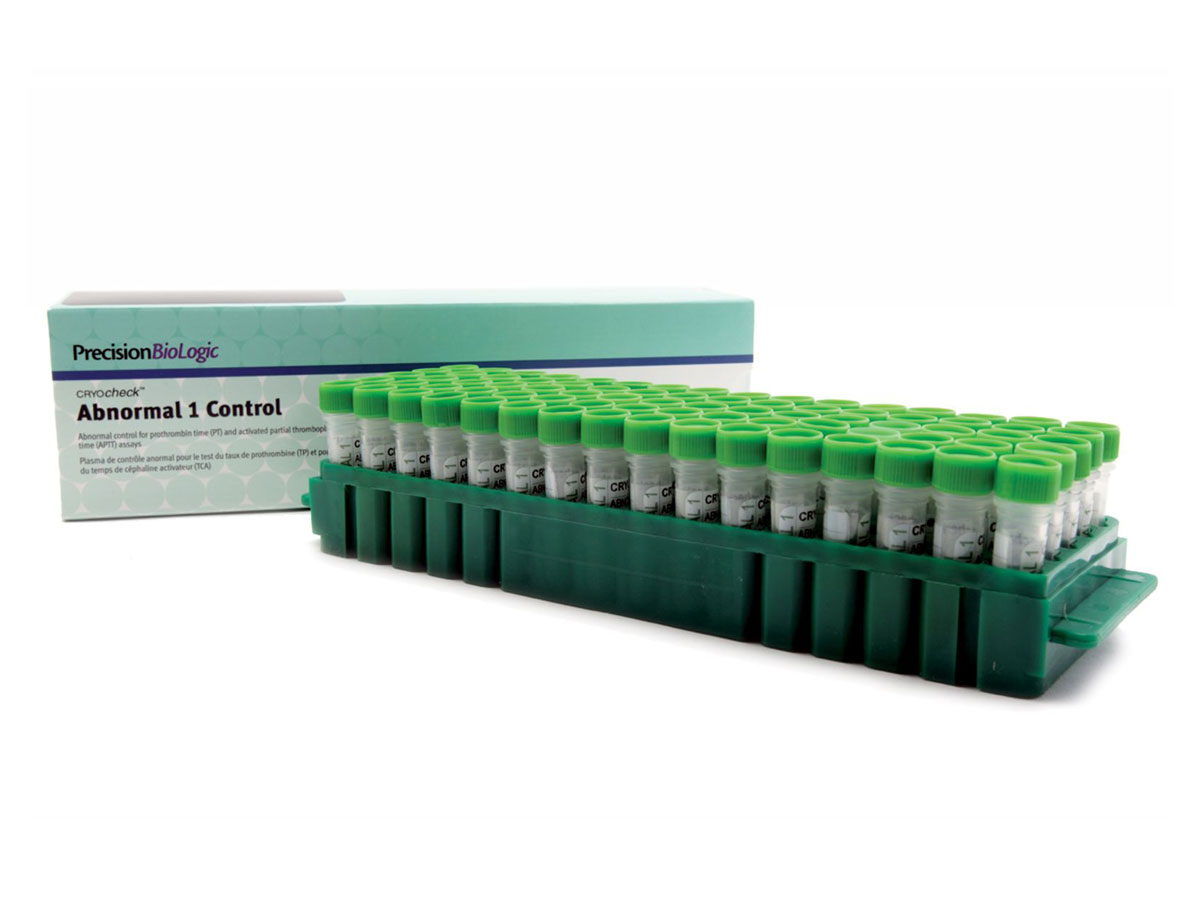
CRYOcheck™ Abnormal 1 Control







Fresh frozen plasmas
Level 1 pathological control plasma.
This routine quality control is titrated for routine hemostasis tests (QT, PT, aPTT, Fibrinogen).
Access publications on the manufacturer's website
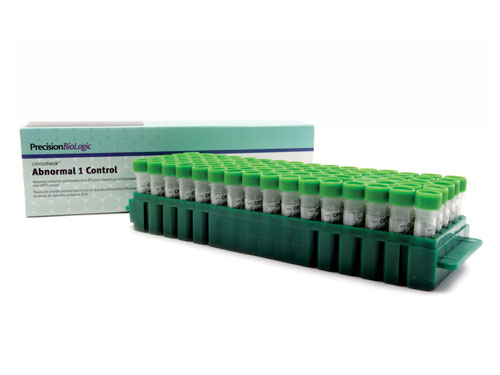
| Reference | CCA1-10 |
|---|---|
| Presentation | Kit |
| Format | 80 x 1.0 mL |
| Quote |
Price list, safety data sheets and notices are accessible to our registered customers.
| Reference | Presentation | Format | Quote | Product sheet |
|---|---|---|---|---|
| CCA1-10 | Kit | 80 x 1.0 mL |