HEMOSTASIS COAGULATION ROUTINE FIBRINOLYSIS TISSUE PLASMINOGEN ACTIVATOR ANTIGEN
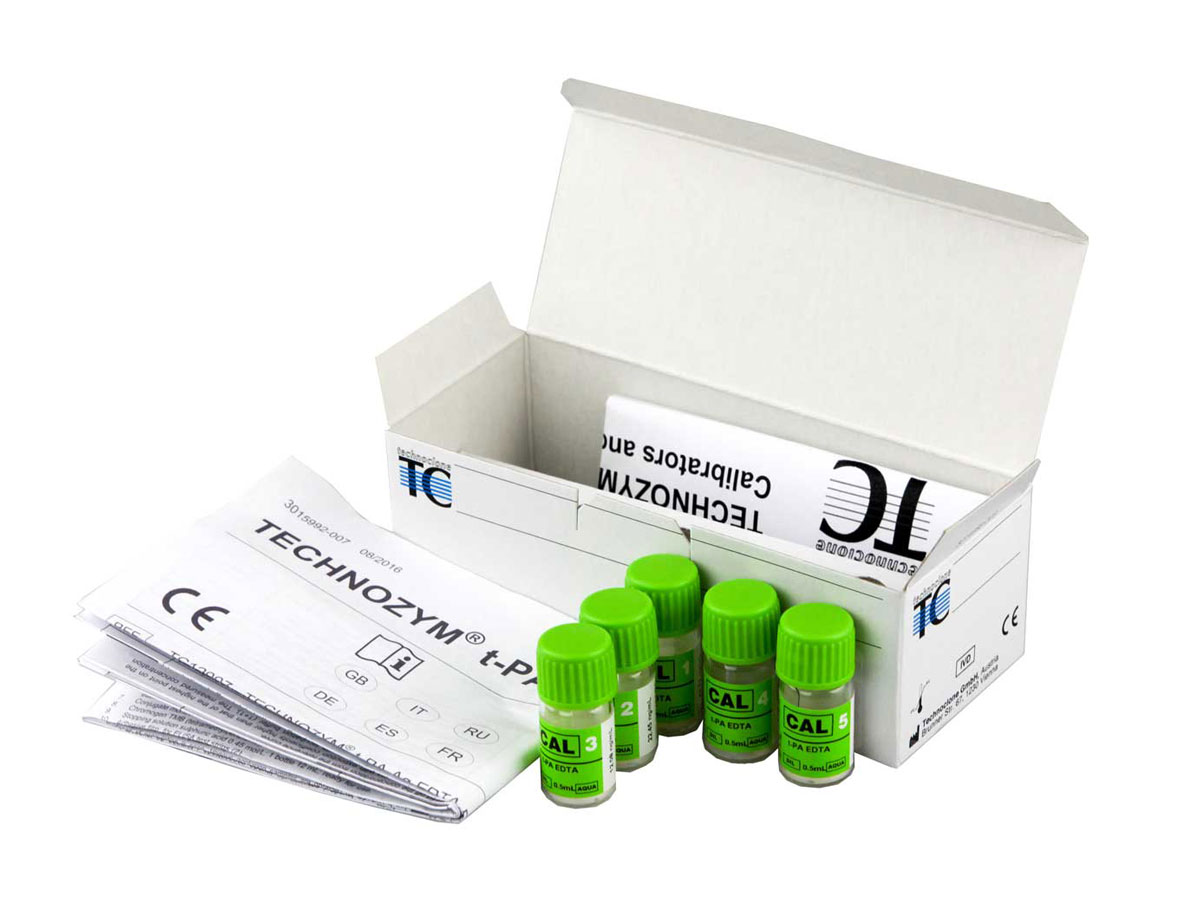
TECHNOZYM® t-PA Calibrator Set








ELISA Assay
Additional calibration plasmas for the antigenic assay of t-PA.
A range of 5 additional calibrators for the TECHNOZYM® t-PA antigen kit.
Access on the manufacturer's website TECHNOCLONE
| Reference | 4-TC12001 |
|---|---|
| Presentation | Vial |
| Format | 5 x 0.5 mL |
| Quote |
Price list, safety data sheets and notices are accessible to our registered customers.
| Reference | Presentation | Format | Quote | Product sheet |
|---|---|---|---|---|
| 4-TC12001 | Vial | 5 x 0.5 mL |