HEMOSTASIS COAGULATION ROUTINE FACTOR ASSAYS CHROMOGENIC ASSAYS TAFI
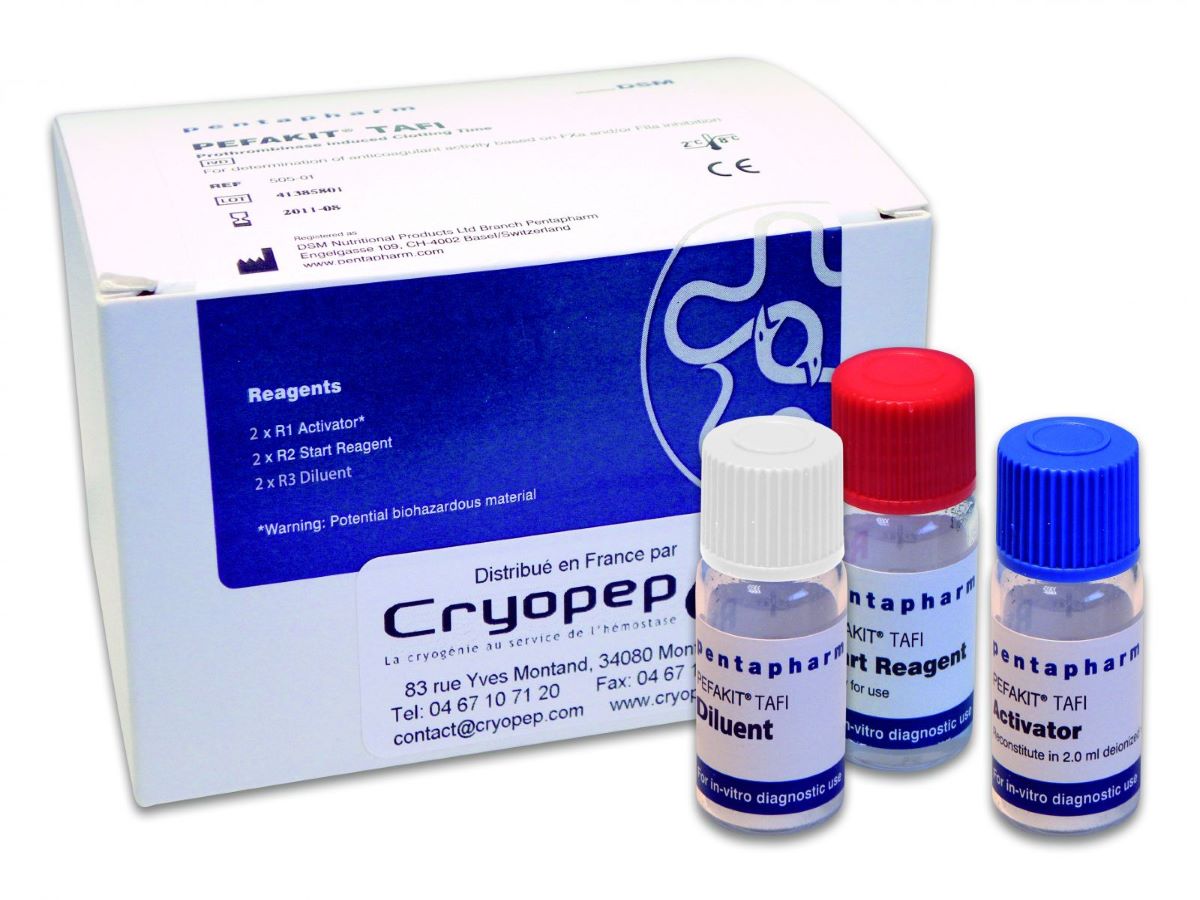
Pefakit® TAFI








Pefakit® TAFI is a plasma-based chromogenic test for the determination of TAFI activity.
| Reference | 8-800186 |
|---|---|
| Presentation | Kit |
| Format | 2 x 4.0 mL |
| Quote |
Price list, safety data sheets and notices are accessible to our registered customers.
| Reference | Presentation | Format | Quote | Product sheet |
|---|---|---|---|---|
| 8-800186 | Kit | 2 x 4.0 mL |