HEMOSTASIS COAGULATION ROUTINE LUPUS DIAGNOSTICS (LA) POSITIVE CONTROL
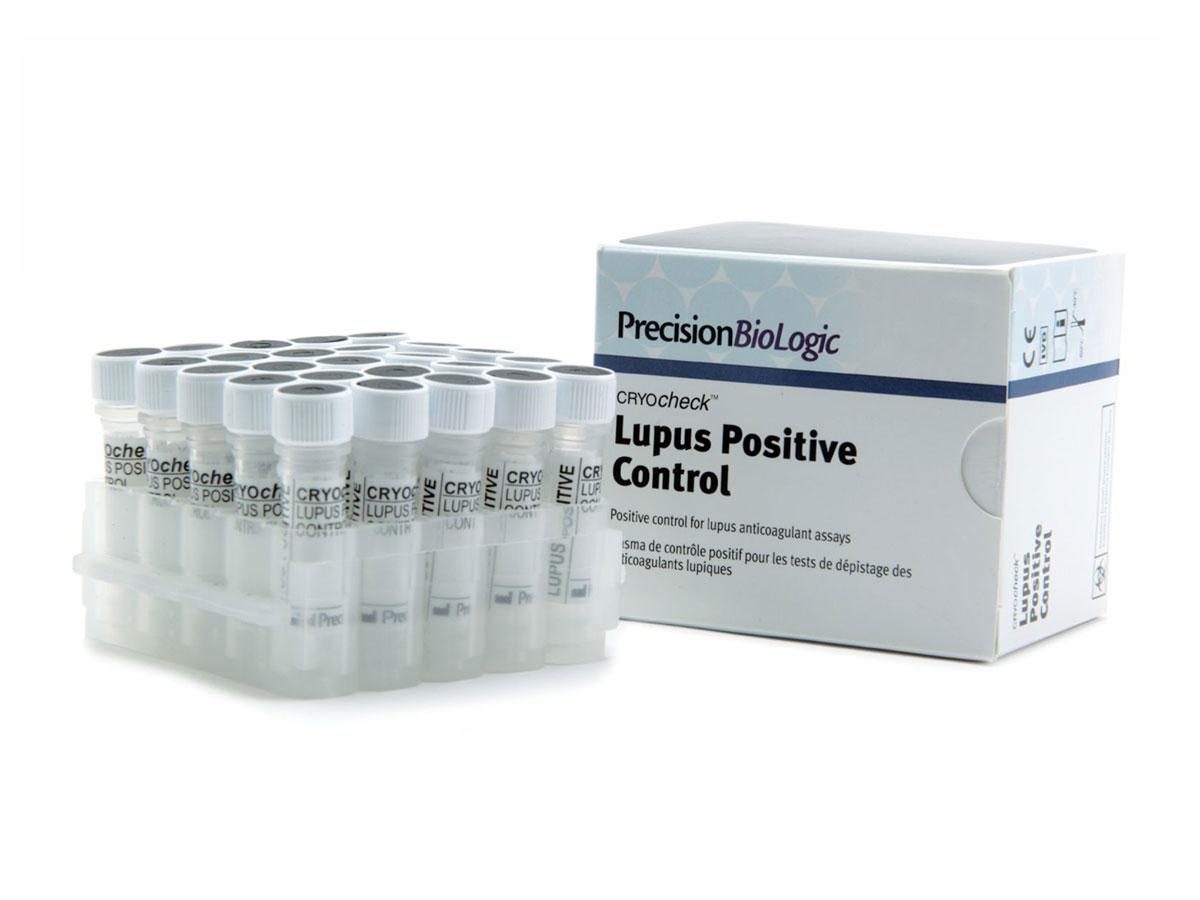
CRYOcheck™ Lupus Positive Control




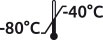


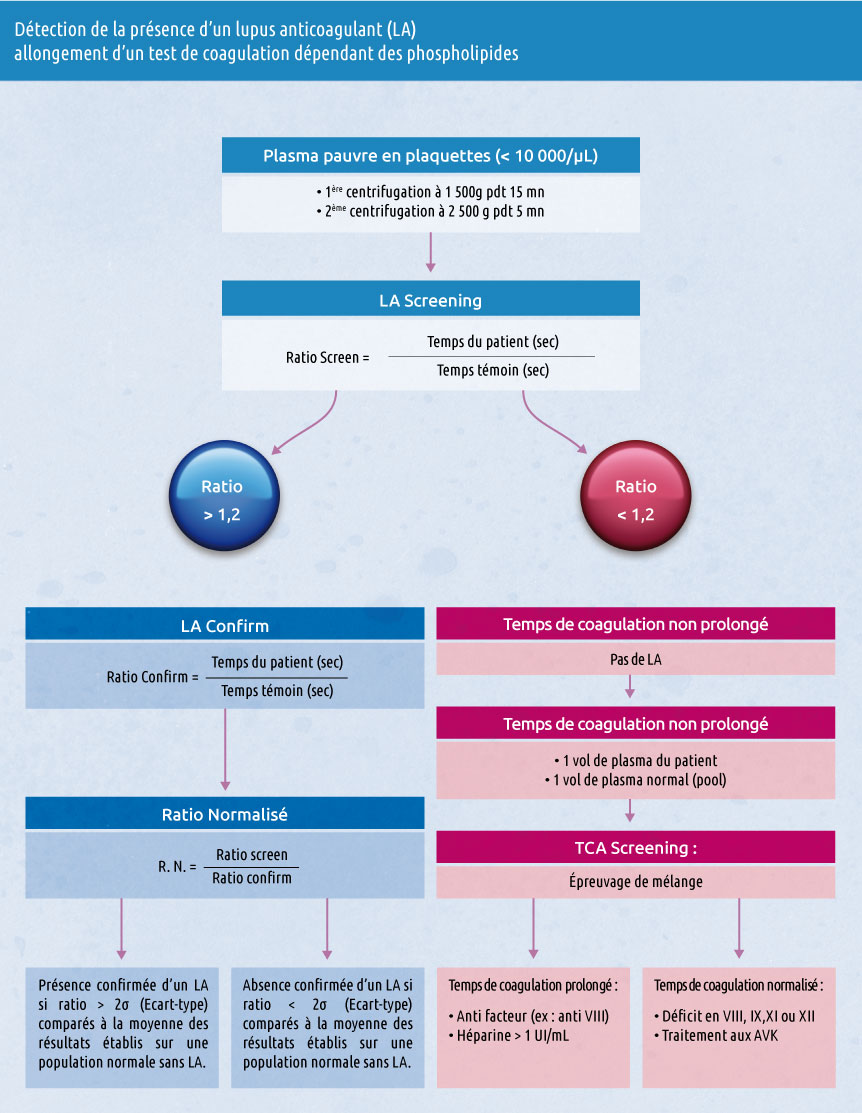
Chronometric assay
Strong positive plasma for lupus anticoagulant (LA) assays.
CRYOcheck™Lupus Positive Control plasma is prepared from plasmas of patients with anticoagulant lupus. It is therefore recommended as a strong positive control for LA detection tests.
Access publications on the manufacturer's website
| Reference | CCLP-05 |
|---|---|
| Presentation | Kit |
| Format | 25 x 0.5 mL |
| Quote |
| Reference | CCLP-10 |
|---|---|
| Presentation | Kit |
| Format | 25 x 1.0 mL |
| Quote |
Price list, safety data sheets and notices are accessible to our registered customers.
| Reference | Presentation | Format | Quote | Product sheet |
|---|---|---|---|---|
| CCLP-05 | Kit | 25 x 0.5 mL | ||
| CCLP-10 | Kit | 25 x 1.0 mL |